After White House weighs in, SF doctor defends decision to offer booster shot for J&J recipients


SAN FRANCISCO (KGO) -- A decision about booster shots in San Francisco has Washington's attention. After ABC7's story aired Monday night, the CDC, FDA, and White House all had something to say about San Francisco's decision to offer a supplemental Pfizer or Moderna shot to people who got the single-dose Johnson and Johnson vaccine.
RELATED: SF General Hospital to give supplemental mRNA vaccine to people who got J&J shot
When asked if the Biden administration endorsed San Francisco's decision at a press briefing, White House press secretary, Jen Psaki said "That's not based on public health, current public health guidance provided by the CDC, that has not changed again. We again, know different localities will be making decisions, but that's not reflective of current public health guidance."
The CDC and FDA also issued statements:
CDC: "People who are fully vaccinated with any of the FDA-authorized vaccines are protected from severe disease and death, including from the variants currently circulating in the country such as Delta. CDC recommends either getting one dose of Johnson & Johnson's COVID-19 vaccine or two doses of Pfizer or Moderna's COVID-19 vaccine to protect you from COVID-19. At this time, CDC does not recommend additional doses or booster shots. Additionally, vaccine providers should continue to administer vaccine in accordance with the current EUA per the COVID-19 vaccine provider agreement."
FDA: "COVID vaccines authorized for emergency use by FDA are authorized based on data and information that support the vaccines' use in specific populations. Use of a COVID vaccine outside the scope of the specified conditions of use in the emergency use authorization (EUA) would not be considered authorized under the EUA."
VIDEO: 'Just wait': Health officials respond to self-prescribed COVID-19 booster shots
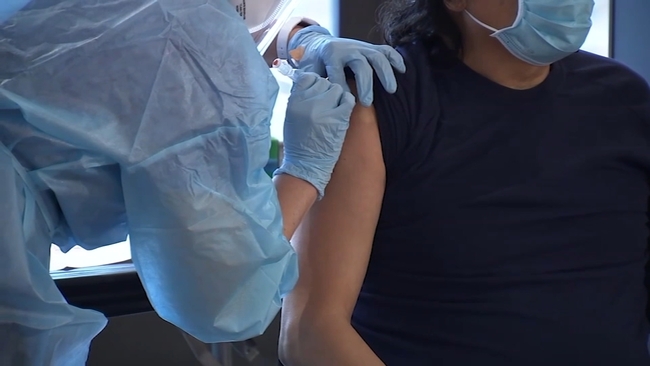
"I was a little surprised at the amount of attention it got only because this has been a discussion for a lot of people for a while," said San Francisco General's chief of emergency medicine, Dr. Chris Colwell, who first shared the news about the extra mRNA vaccine on ABC7.
In a press conference, San Francisco health officials were clear - they're not recommending the extra dose, but rather accommodating special requests.
"In terms of a policy, the recommendation was to accommodate these individuals. How each individual entity does this is according to them," said Dr. Naveena Bobba, Deputy Director of Health of SFDPH.
Dr. Chris Colwell: "I'm very supportive of San Francisco General's decision to allow people who got the J&J shot to get one of the mRNA shots as a booster."
Kate Larsen: "Despite what the FDA, CDC, and White House said today?"
Dr. Chris Colwell: "And I don't necessarily disagree with what they're saying. They're saying there isn't data to say you must do this, I agree with that. I'm not saying you need to go do this. I'm saying if it's something you want to do, you should be able to do it."
Despite the controversy, New York City's health department emailed Dr. Colwell and SFGH chief of staff, Dr. Lisa Winston, citing ABC7's report. The email, which ABC7 news reporter Kate Larsen obtained, says New York had "similar discussions" and wanted to know how the hospital came to its decision and addressed concerns about liability.
VACCINE TRACKER: Here's how CA is doing, when you can get a coronavirus vaccine
"The New York Department of Public Health is one of many that has considered something like this and tried to balance this very issue that we're dealing with. The FDA isn't recommending it, but on the other hand does that mean that we shouldn't do it?" asked Dr. Colwell, who continued, "If the evidence that we have suggests it may help, and it won't hurt, then doesn't it make sense to consider this?"
The extra shots are expected to be available at San Francisco General Hospital in the next week.
Having trouble loading the tracker above? Click here to open it in a new window. RELATED STORIES & VIDEOS:
- Map shows which counties can, can't reopen under reopening tiers
- Cheat sheet: What you can and can't do after being fully vaccinated
- How to register for a COVID-19 vaccine in every Bay Area county
- Map shows everywhere you can get a COVID-19 test in the Bay Area
- Interactive map shows what's closed and what's reopening in the San Francisco Bay Area
- Data tracker: Coronavirus cases, deaths, hospitalizations in every Bay Area county
- Third stimulus check calculator: See how much you could get
- COVID-19 Diaries: Personal stories of Bay Area residents during pandemic
- Get the latest updates on California EDD, stimulus checks, unemployment benefits
- Coronavirus origin: Where did COVID-19 come from?
- What is a COVID-19 genetic, antigen and antibody test?
- What does COVID-19 do to your body and why does it spread so easily?
- Coronavirus Timeline: Tracking major moments of COVID-19 pandemic in San Francisco Bay Area
- Coronavirus Doctor's Note: Dr. Alok Patel gives his insight into COVID-19 pandemic










